



1 / 5










| Still deciding? Get samples of $ ! US$ 324/pcs Request Sample |

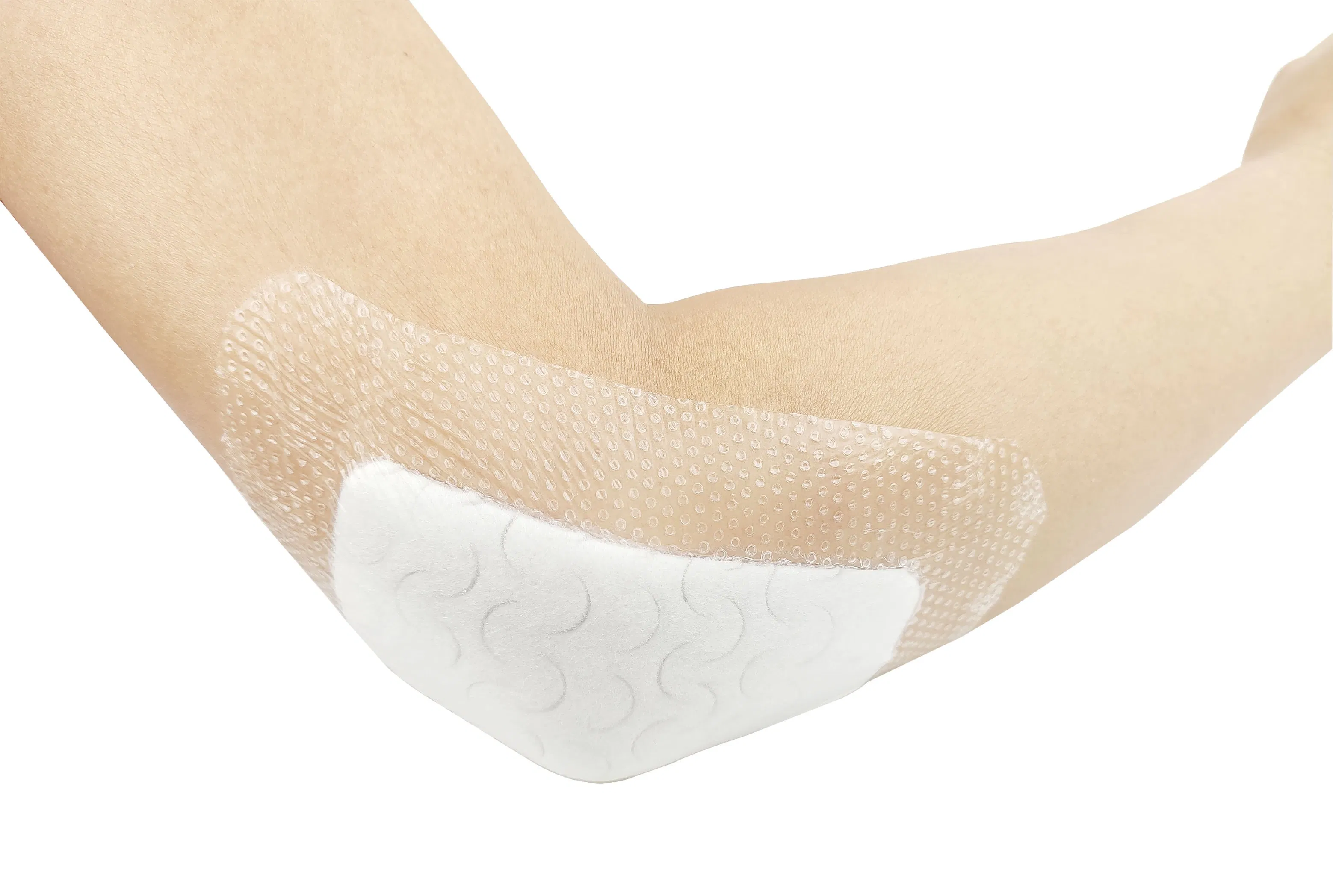
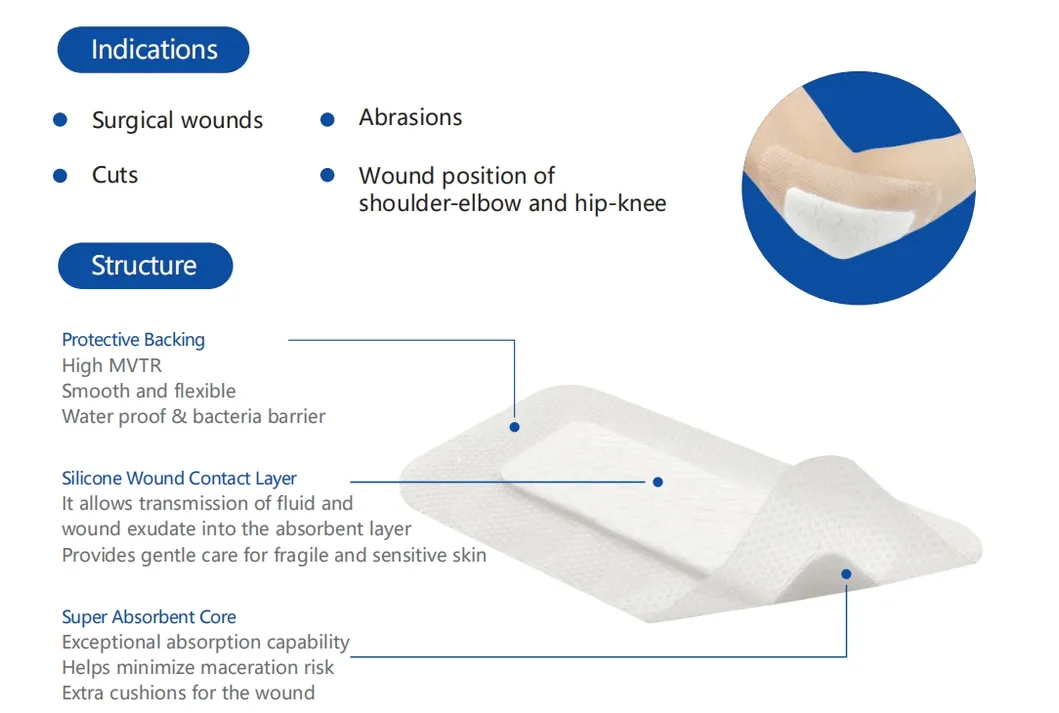
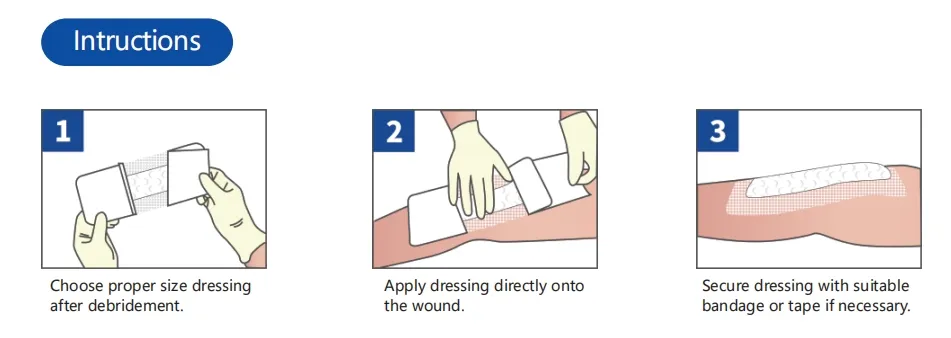
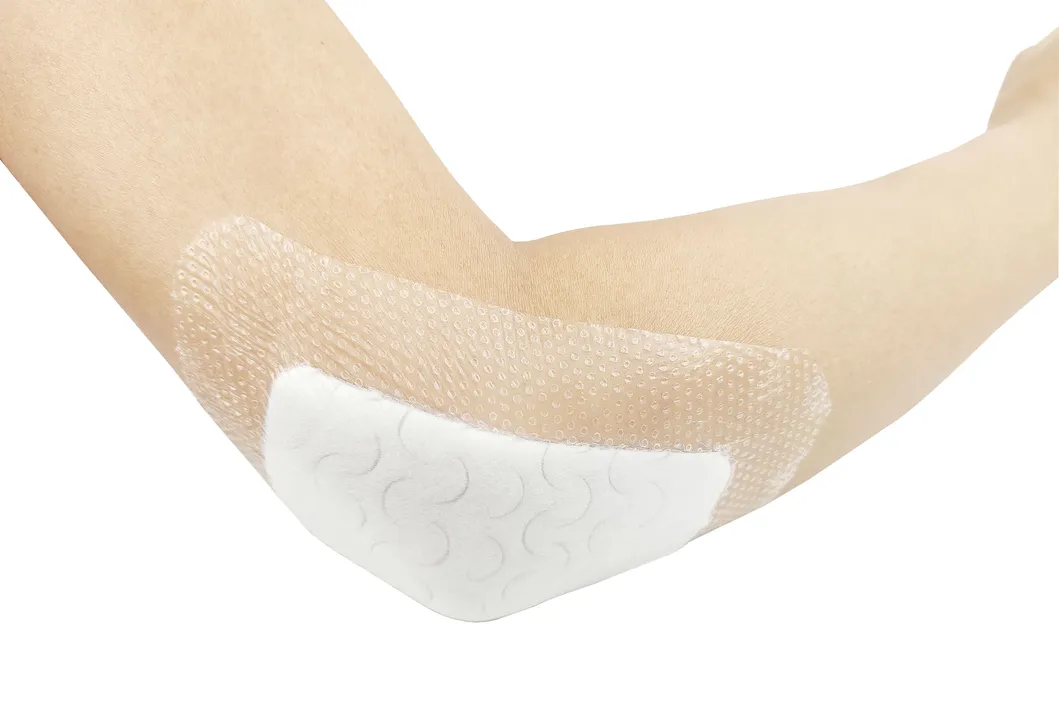
Indicated for management of moderately to heavily exuding wounds, including:


| Model | Specifications | Piece/Box | Boxes/Carton |
|---|---|---|---|
| QxO806 | 8cm*6cm | 10 | 100 |
| Qx1010 | 10cm*10cm | 10 | 100 |
| Qx1510 | 15cm*10cm | 10 | 100 |
| Qx2010 | 20cm*10cm | 10 | 100 |
| Qx2510 | 25cm*10cm | 10 | 100 |
| QX3010 | 30cm*10cm | 10 | 100 |
| Qx3510 | 35cm*10cm | 10 | 100 |






 Seeker Medical
Seeker Medical