












| Still deciding? Get samples of $ ! US$ 0.6/pcs Request Sample |

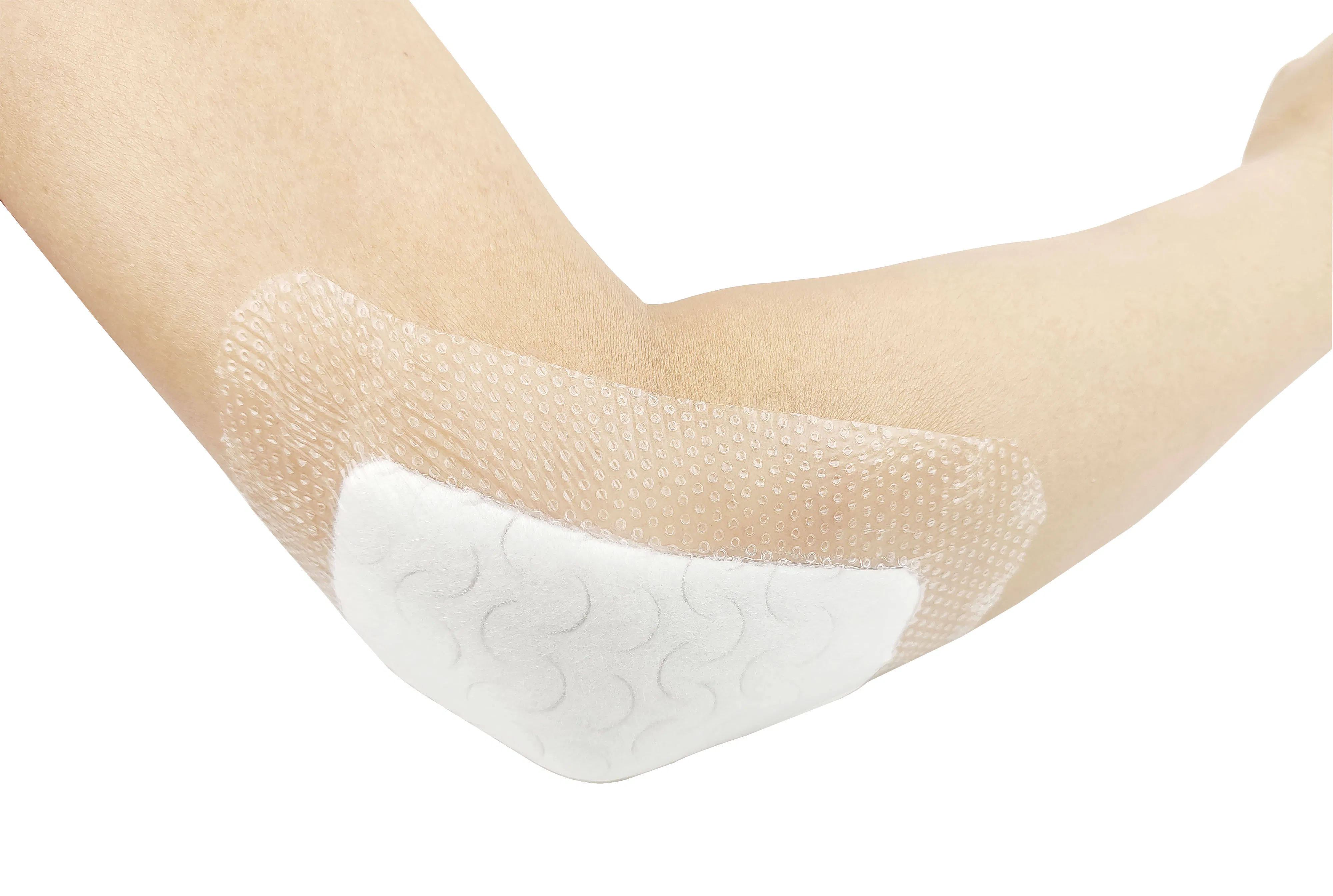
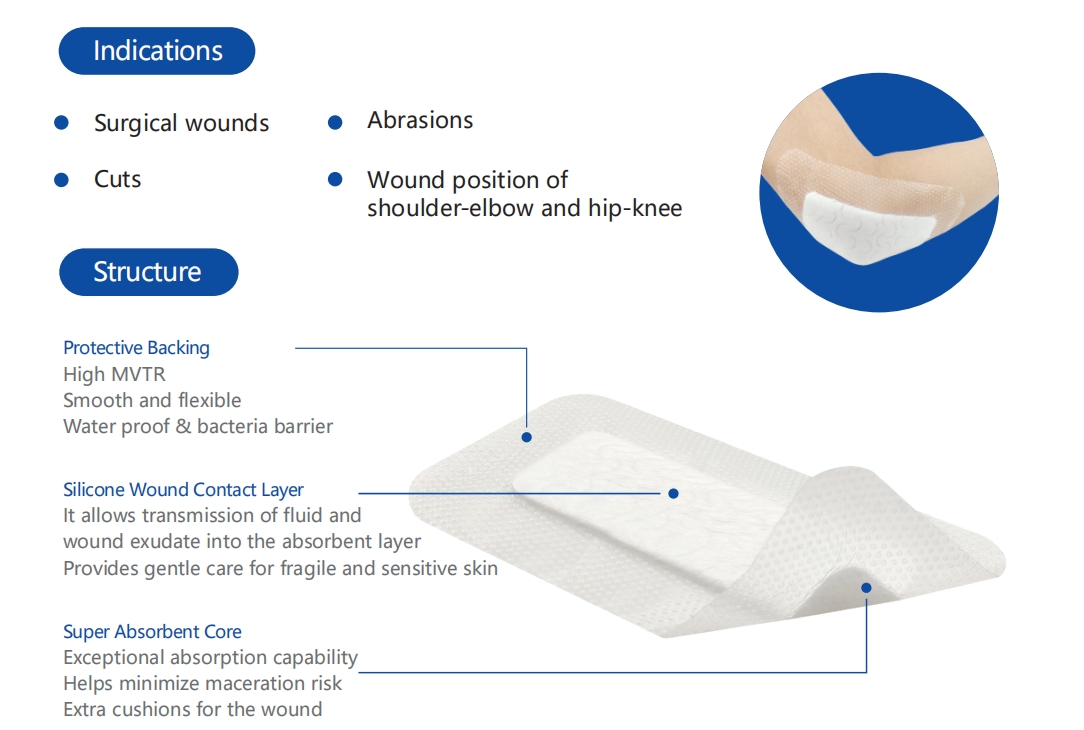
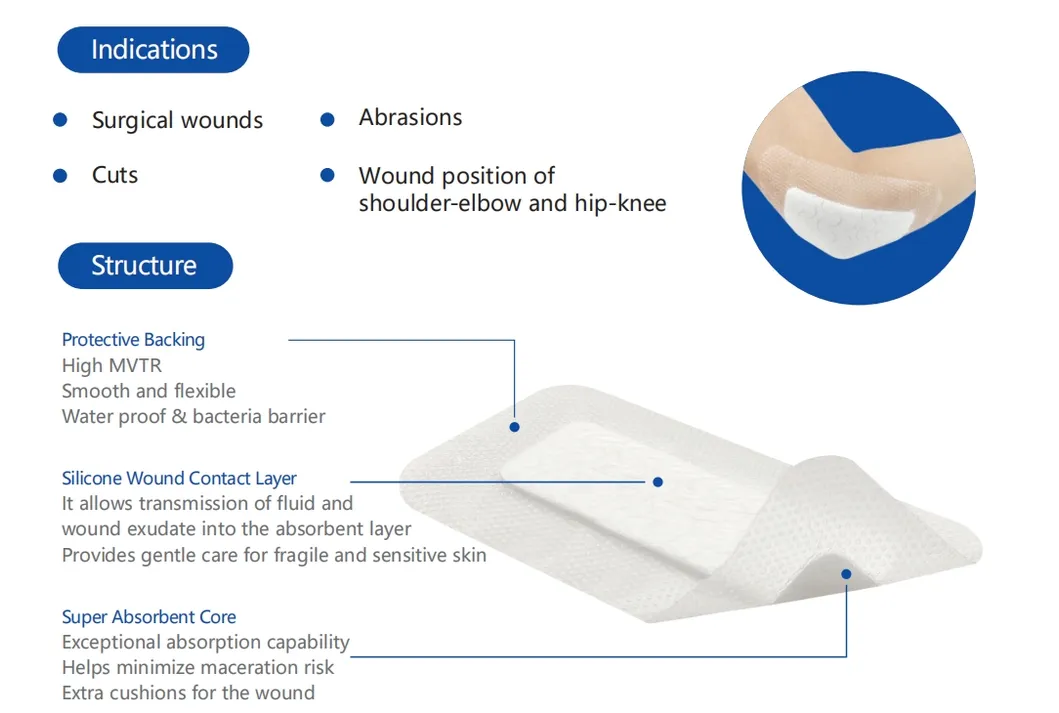
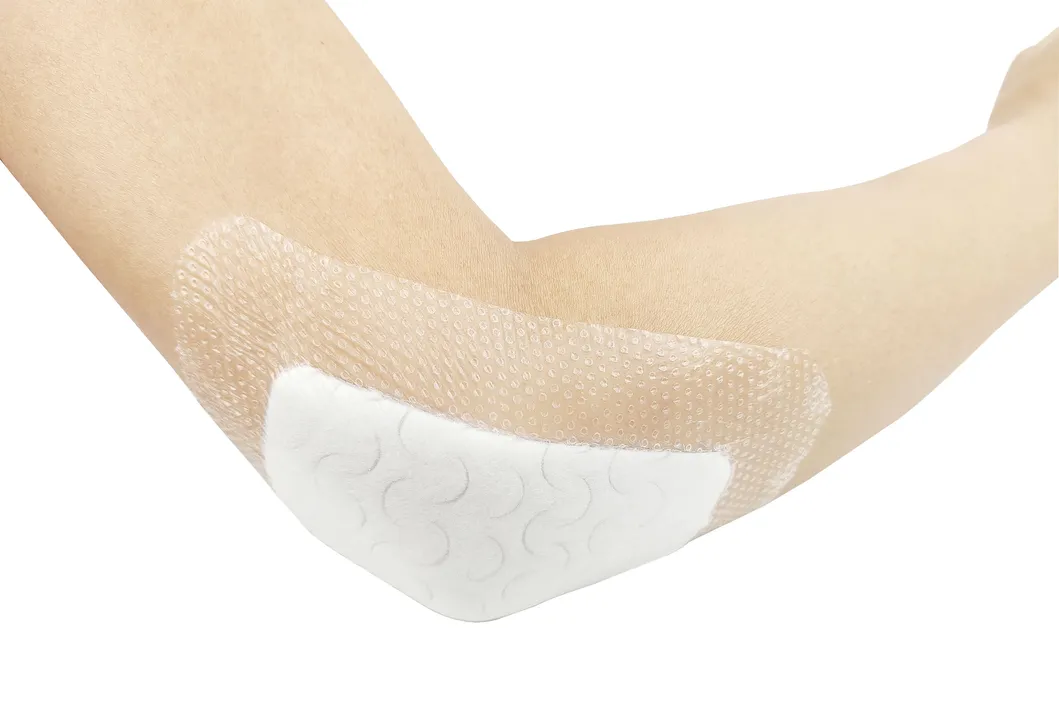
The silicone Post-Operation dressing is composed of a soft silicone wound contact layer, non-woven absorbent layer, and a waterproof breathable film. It is specifically designed for surgical wounds, abrasions, and general trauma. The soft silicone layer ensures firm yet gentle adhesion, minimizing pain and secondary injury during dressing changes.
Indicated for management of moderately to heavily exuding wounds, including: leg ulcers, pressure ulcers, diabetic foot ulcers, and dehisced surgical wounds.


| Model | Specifications | Piece/Box | Total Pcs/Carton |
|---|---|---|---|
| QxO806 | 8cm * 6cm | 10 | 100 |
| Qx1010 | 10cm * 10cm | 10 | 100 |
| Qx1510 | 15cm * 10cm | 10 | 100 |
| Qx2010 | 20cm * 10cm | 10 | 100 |
| Qx2510 | 25cm * 10cm | 10 | 100 |
| QX3010 | 30cm * 10cm | 10 | 100 |
| Qx3510 | 35cm * 10cm | 10 | 100 |


Specializing in medical technology since 2015, our facility utilizes advanced production equipment to deliver high-quality medical surface catheter fixation devices, transparent dressings, and fixation bands. We operate under a rigorous scientific management system, holding qualifications for both Class I and Class II medical devices.




 Seeker Medical
Seeker Medical