



1 / 5










| Still deciding? Get samples of $ ! US$ 130/Set Request Sample |
| Product Code | Product Name | Model | Description |
|---|---|---|---|
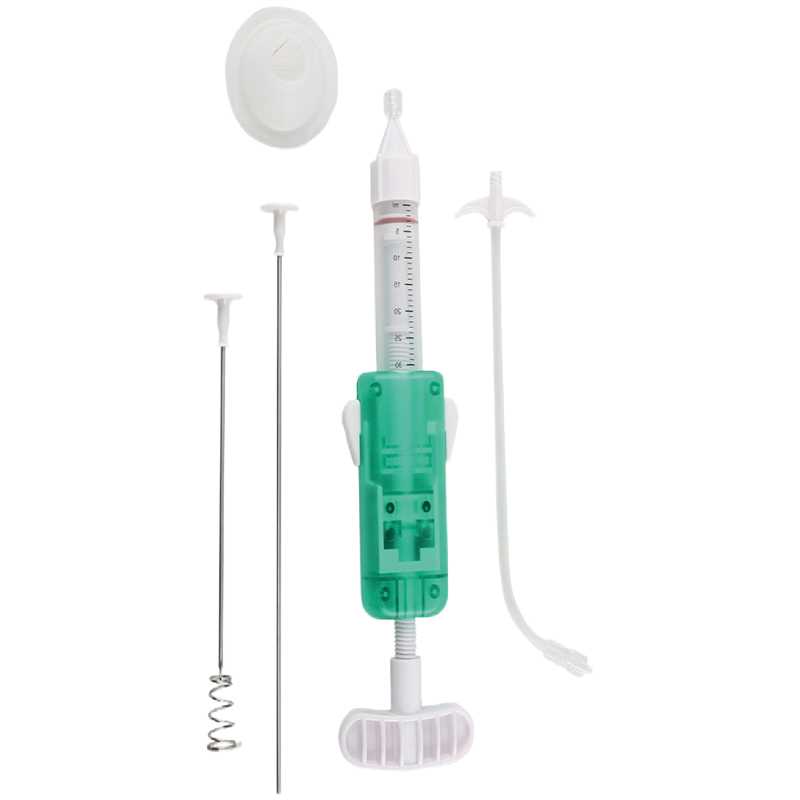
| 030020003 | Kyphoplasty Tool Kit | Type B (Lumbar Spine) | φ3.0 Percutaneous Access Device (1pc), φ3.5 Aiguille (1pc), Guide Wire (1pc), φ4.2 Expansion Cannula (1pc), φ3.5 Bone Cement Applier (4pc) |
| 030020002 | Kyphoplasty Tool Kit | Type D (Lumbar Spine) | φ3.0 Percutaneous Access Device (2pc), φ3.5 Aiguille (1pc), Guide Wire (2pc), φ4.2 Expansion Cannula (2pc), φ3.5 Bone Cement Applier (6pc) |
| 030020001 | Kyphoplasty Tool Kit | Type G (Lumbar Spine) | φ4.2 Percutaneous Access Device (1pc), φ3.5 Aiguille (1pc), Guide Wire (1pc), φ3.5 Bone Cement Applier (4pc) |
| 030030005 | Kyphoplasty Tool Kit | Type T1 (Thoracic Spine) | φ2.4 Percutaneous Access Device (2pc), φ3.0 Aiguille (1pc), Guide Wire (2pc), φ3.6 Expansion Cannula (2pc), φ3.0 Bone Cement Applier (6pc) |
| 030030006 | Kyphoplasty Tool Kit | Type T11 (Thoracic Spine) | φ2.4 Percutaneous Access Device (1pc), φ3.0 Aiguille (1pc), Guide Wire (1pc), φ3.6 Expansion Cannula (1pc), φ3.0 Bone Cement Applier (4pc) |
Our integrated, one-step design allows for quick and efficient percutaneous access to the bone, creating a bone tissue guide channel seamlessly. It effectively reduces trauma during the procedure. Doctors can choose between bevel or diamond tips based on specific clinical requirements.
*All prices are for reference only. Exact pricing is based on the latest formal quotation.





 Seeker Medical
Seeker Medical