Q1: Can I get a sample for testing?
A1: Yes, we can provide samples for free to verify the quality.
Q2: Can I add my logo on the medical products?
A2: Yes, OEM & ODM services are available for customized medical solutions.
Q3: Are the products certified?
A3: Yes, the quality control system has EN ISO 13485 and CE certificates.
Q4: Will you help with registration in my country?
A4: Yes, we will provide all the necessary documents and technical files needed for registration.
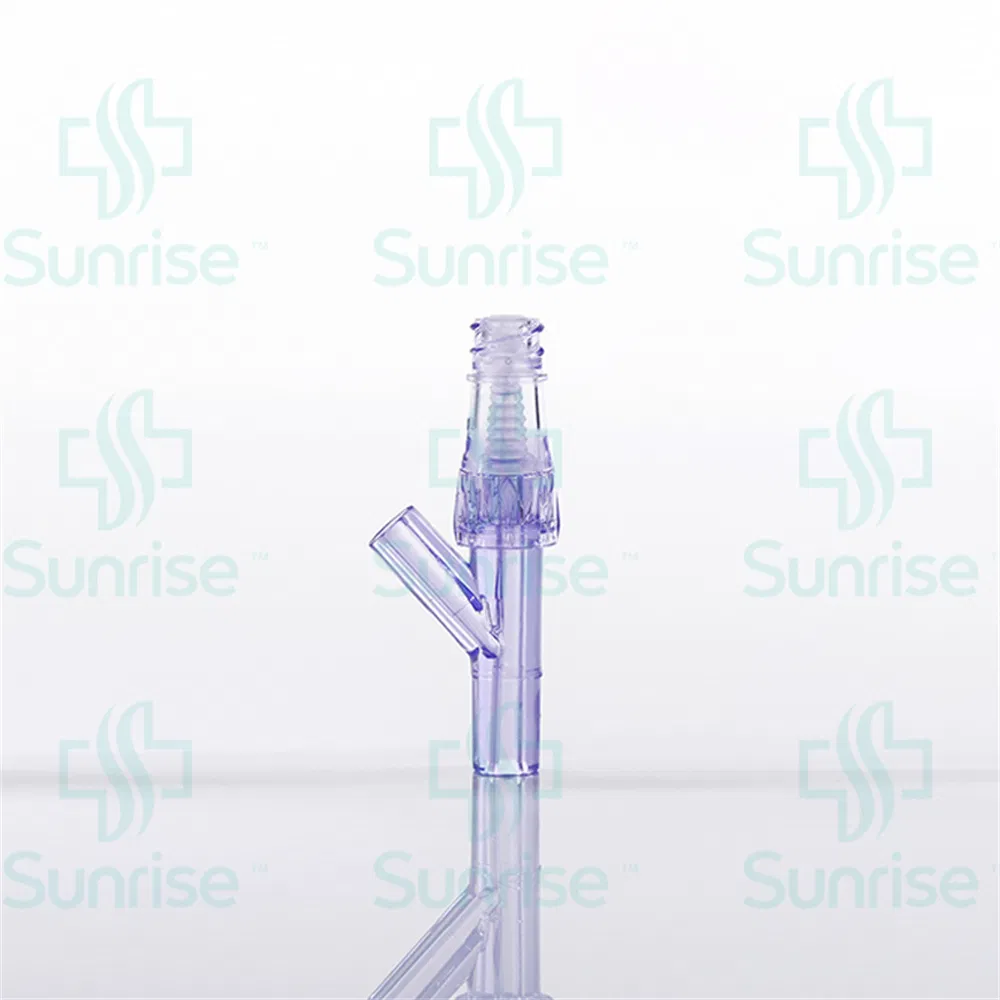
Q5: What is the shelf life of the Needle Free Connector?
A5: The product has a shelf life of 5 years under proper storage conditions.
Q6: What materials are used in the connector?
A6: It is made of high-quality medical grade polycarbonate and medical grade silicone, ensuring safety and compatibility.

















 Seeker Medical
Seeker Medical