


1 / 5










| Still deciding? Get samples of $ ! US$ 0.35/pcs Request Sample |
| Model NO. | A1 |
| Sterilization | Ethylene Oxide Sterilization |
| Quality Guarantee Period | Two Years |
| Group | All Groups |
| Usage | Hospital, Clinic etc |
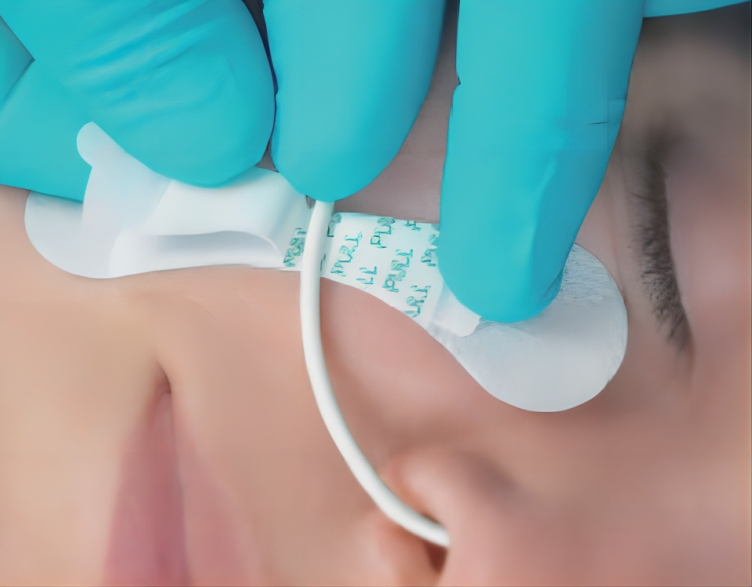
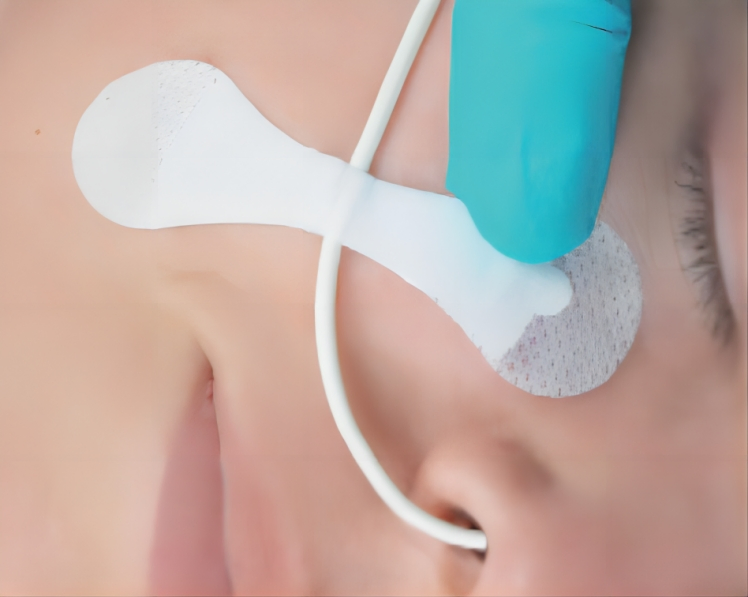
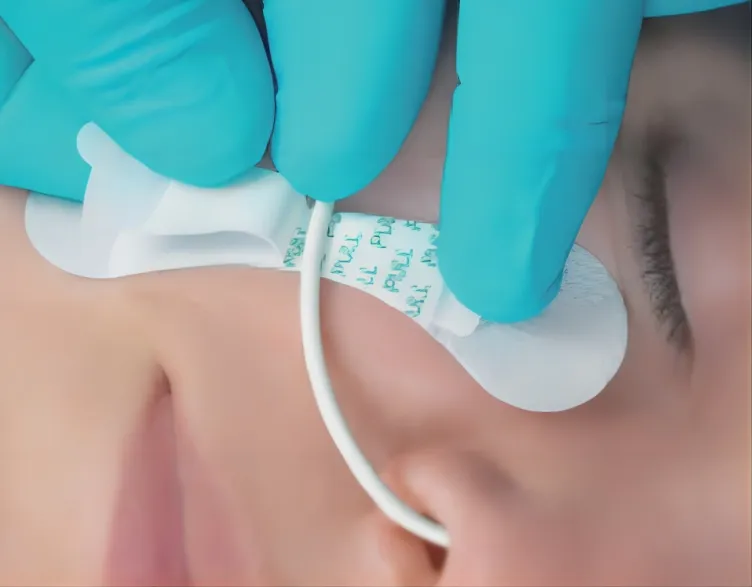
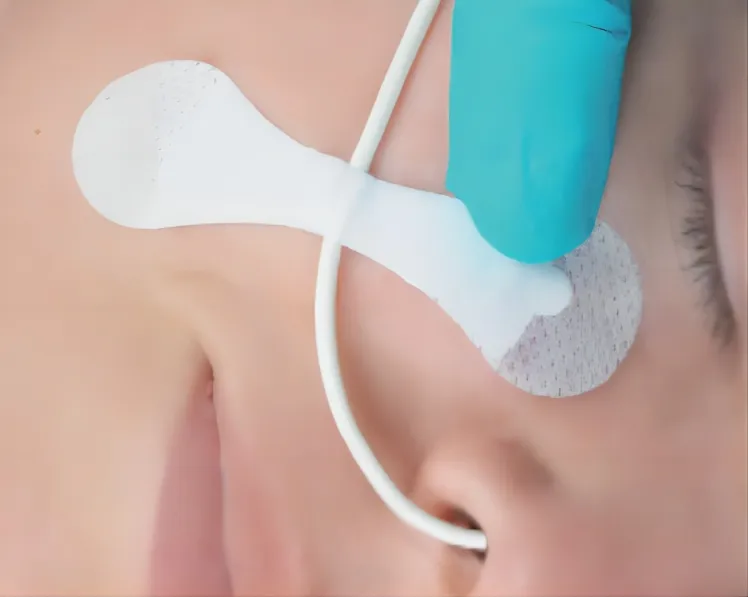
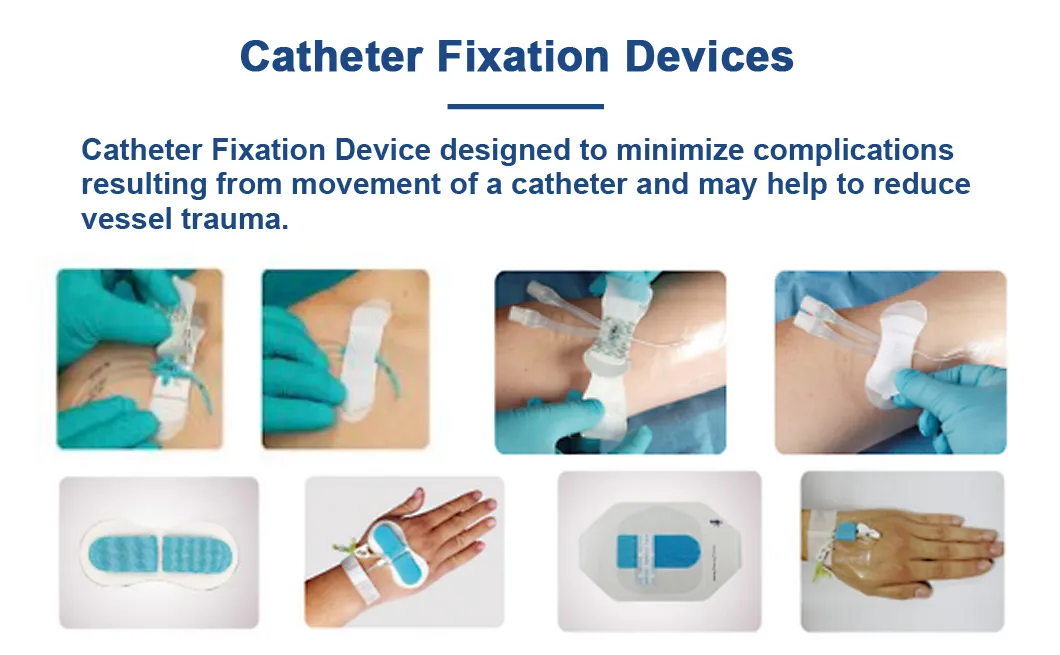
| Application | Reduce Vessel Trauma |
| Product Name | Catheter Fixation Device |
| Certificate | CE, ISO13485 |
| Trademark | TECHDRIVE |
| Production Capacity | 100000 Pieces/Week |

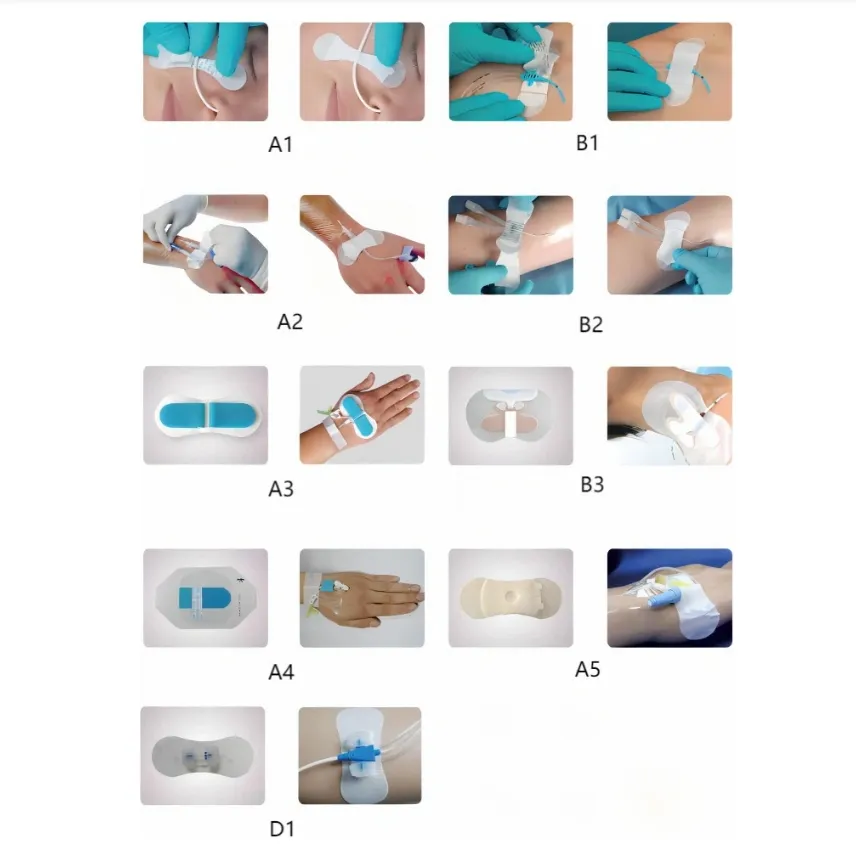
| Type | Code | Pcs/inner box | Pcs/master carton |
|---|---|---|---|
| A | A1 | 50 | 500 |
| A2 | 50 | 500 | |
| A3 | 50 | 500 | |
| A4 | 50 | 500 | |
| A5 | 50 | 500 | |
| B | B1 | 50 | 500 |
| B2 | 50 | 500 | |
| B3 | 50 | 500 | |
| D | D1 | 50 | 500 |
We are a medical technology company approved by relevant health authorities. Founded in 2015 with a focus on high-quality medical consumables, the company invested heavily in advanced production equipment from abroad. With a scientific management system, we have grown into a leading specialist in the industry.
Our expertise lies in the development, production, and sale of medical surface catheter fixation devices, transparent dressings, and fixation bands. We hold production qualifications for Class I and Class II medical devices, ensuring clinical safety and professional reliability.




 Seeker Medical
Seeker Medical