













| Still deciding? Get samples of $ ! US$ 0.36/pcs Request Sample |

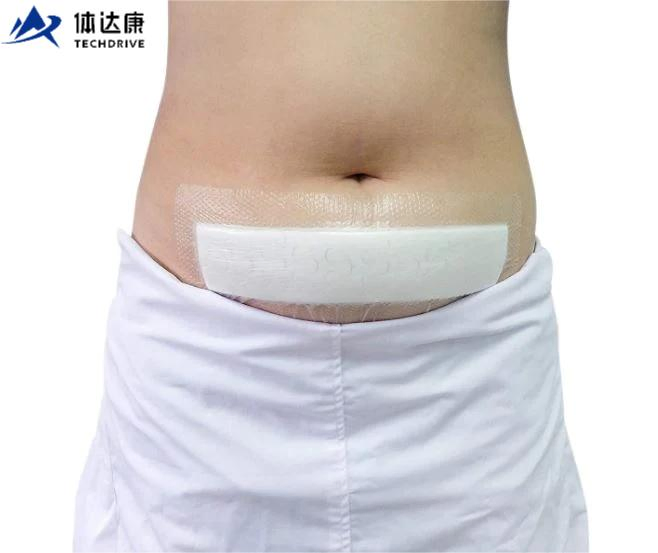
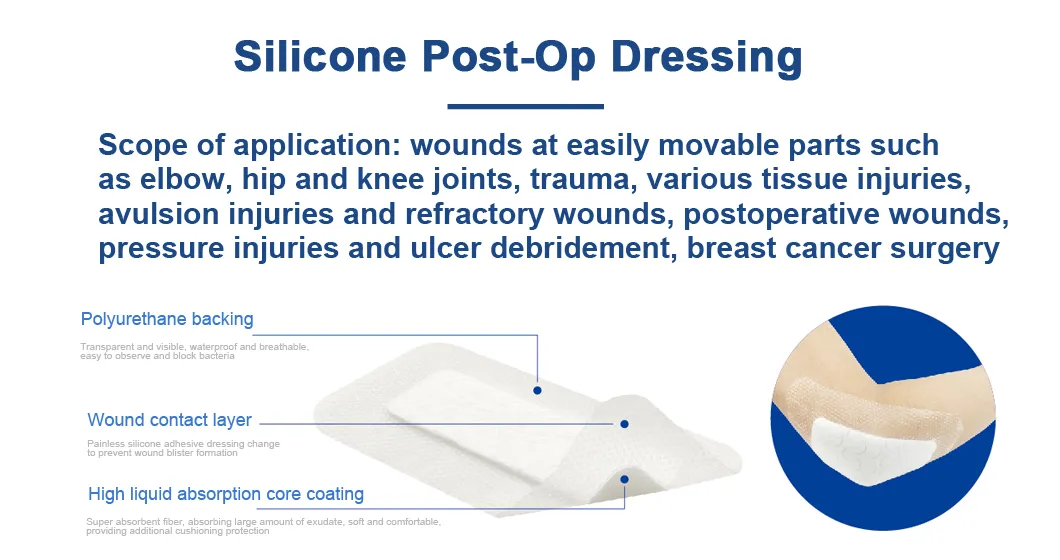
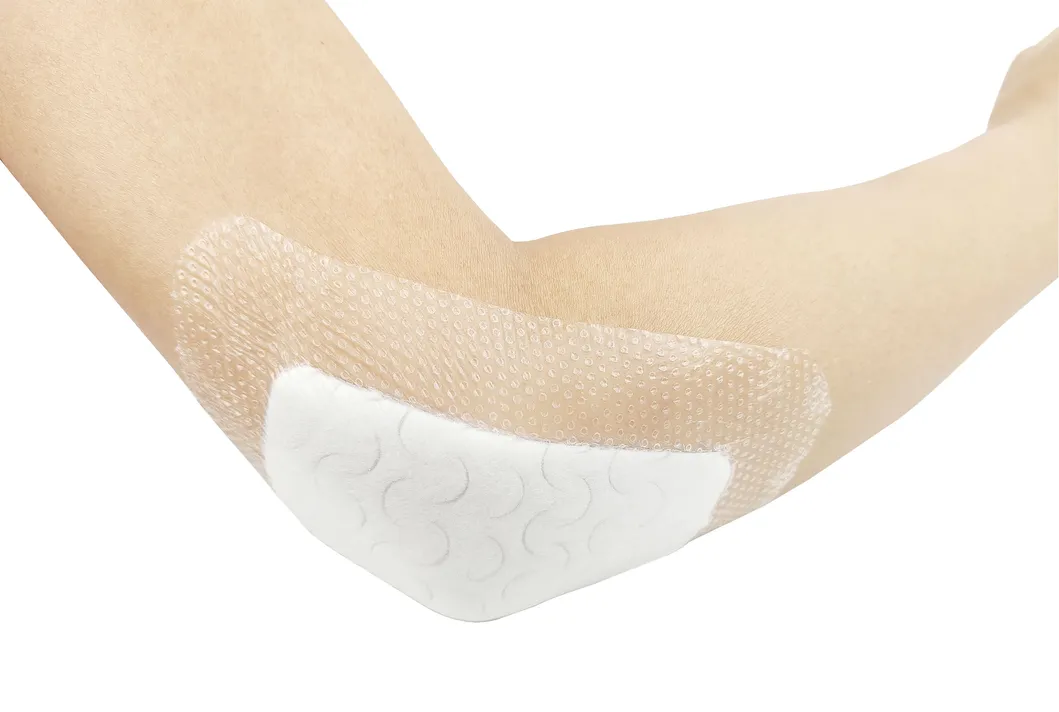
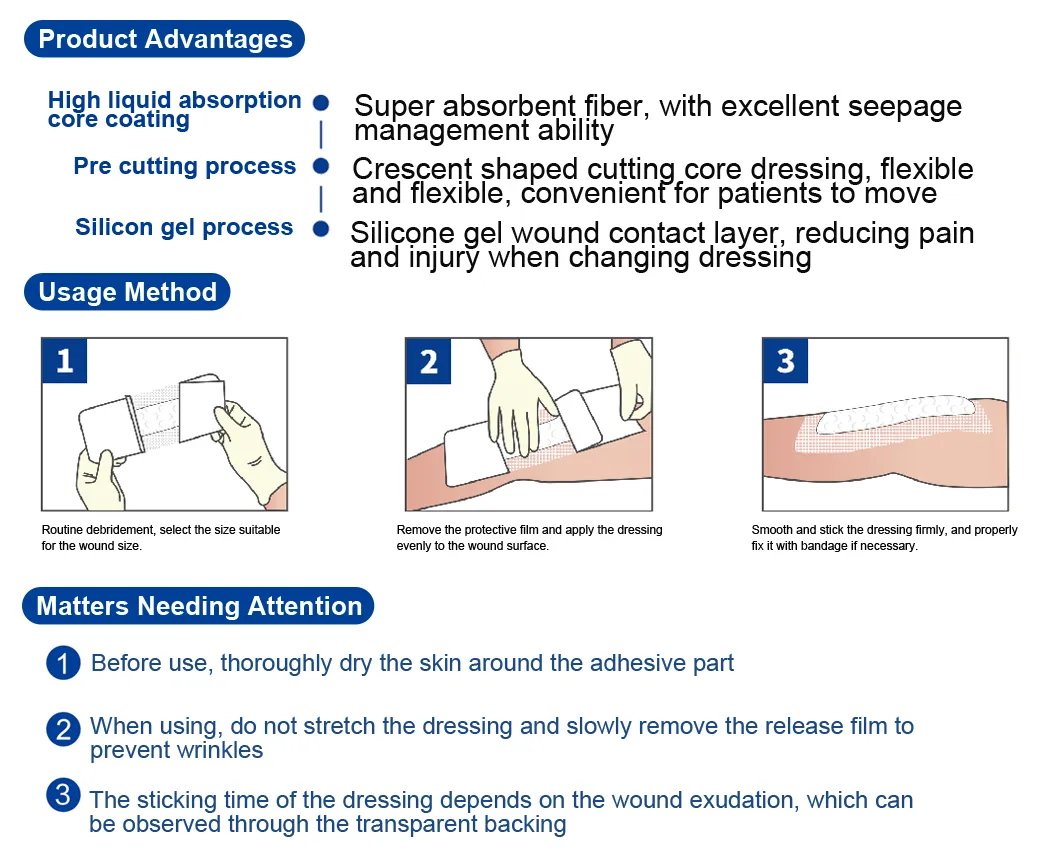
The silicone Post-Operation dressing is composed of a soft silicone wound contact layer, non-woven absorbent layer and a waterproof breathable film, which is suitable for wounds and abrasions particularly for surgical wounds. The silicone wound contact layer is soft and firm which can minimize the pain of changing the dressing.
Silicone Post-operative Dressing is indicated for management of moderately to heavily exuding wounds, including: leg ulcers, pressure ulcers, diabetic foot ulcers and dehisced surgical wounds.

| Model | Specifications | Piece/box | Pcs./carton |
|---|---|---|---|
| Qx0806 | 8cm*6cm | 10 | 100 |
| Qx1010 | 10cm*10cm | 10 | 100 |
| Qx1510 | 15cm*10cm | 10 | 100 |
| Qx2010 | 20cm*10cm | 10 | 100 |
| Qx2510 | 25cm*10cm | 10 | 100 |
| QX3010 | 30cm*10cm | 10 | 100 |
| Qx3510 | 35cm*10cm | 10 | 100 |


Our facility is a medical technology company approved by provincial government and registered by Food and Drug Administration. Founded in 2015, we have introduced advanced production equipment and maintain a perfect and scientific management system. We specialize in the development, production, and sale of medical surface catheter fixation devices, transparent dressings, and fixation bands. We hold production qualifications for Class I and Class II medical devices.



 Seeker Medical
Seeker Medical