
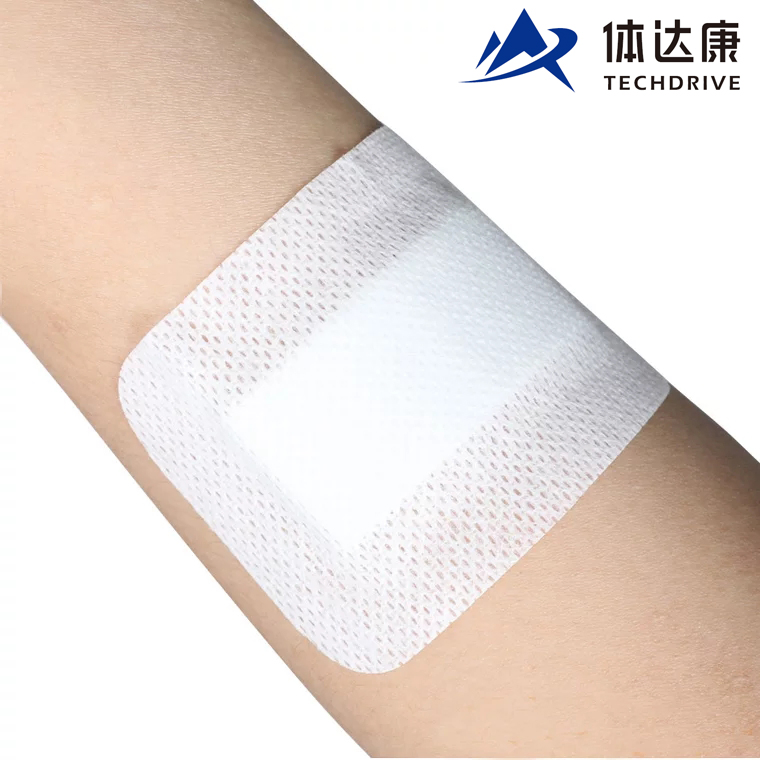



1 / 5










| Still deciding? Get samples of $ ! US$ 0.03/Piece Request Sample |

| Model | Specifications | Dimensions | Sheet/Box |
|---|---|---|---|
| Type A | A-101 / A-103 | 6cm*7cm / 10cm*10cm | 20 |
| A-104 | 10cm*15cm | 20 | |
| A-105 | 10cm*20cm | 20 | |
| A-106 | 10cm*25cm | 20 | |
| A-121 | 6cm*9cm | 20 | |
| A-113 | 9cm*20cm | 20 | |
| Type B1 | B-101 / B-103 | 6cm*7cm / 10cm*12cm | 20 |
| B-105 | 10cm*20cm | 20 | |
| B-114 | 4.4cm*4.4cm | 20 | |
| B-109 | 15cm*15cm | 20 |






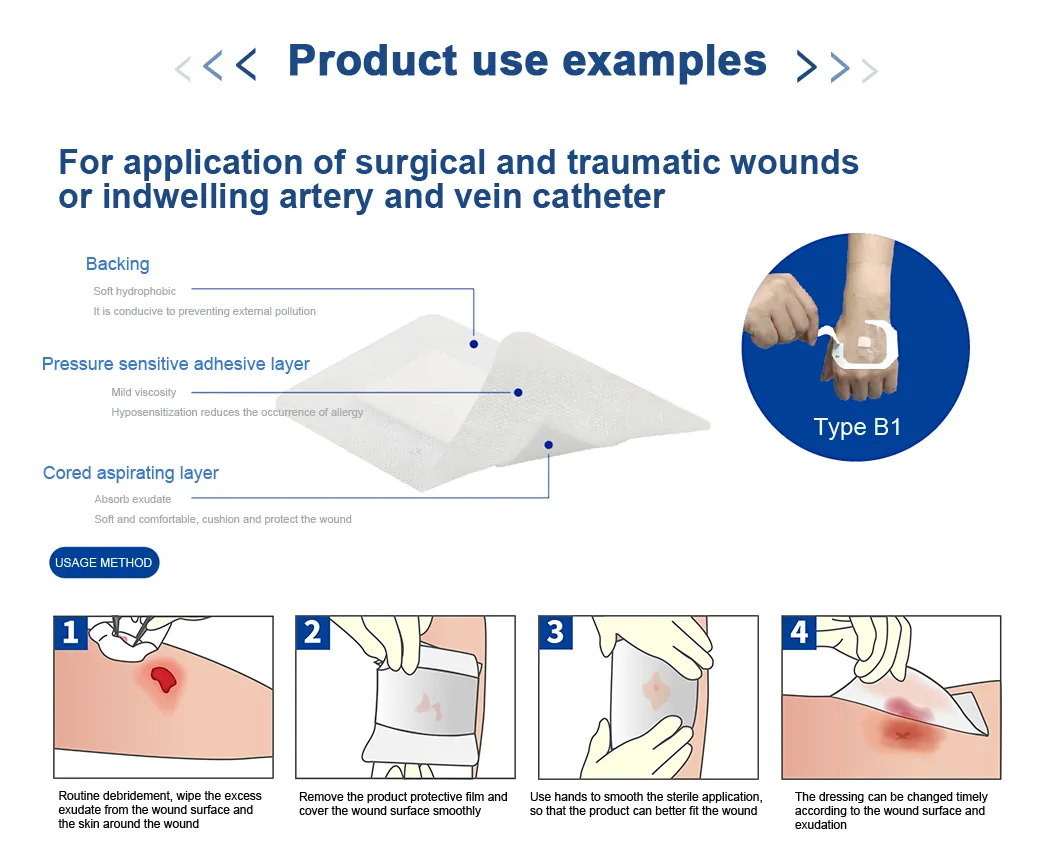

The company is a medical technology company approved by the government and registered by the Food and Drug Administration. Founded in 2015, the company invested more than 6 million yuan to introduce advanced production equipment from abroad, maintaining a perfect and scientific management system. The company has the capability to develop, produce, and sell medical surface catheter fixation devices, transparent stickers, transparent dressings, fixation bands, and other products, holding production qualifications for Class I and Class II medical devices.


 Seeker Medical
Seeker Medical