


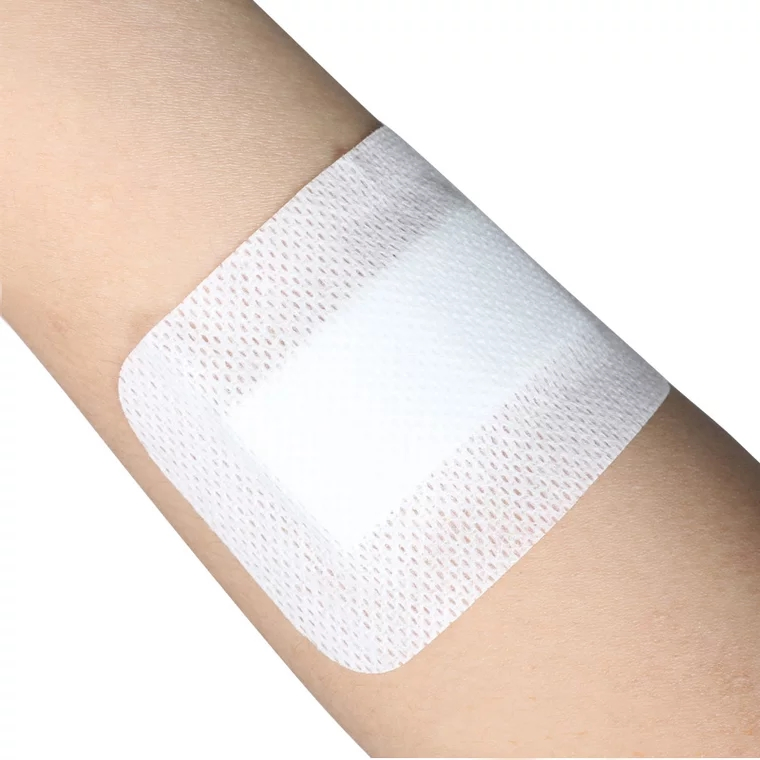

1 / 5










| Still deciding? Get samples of $ ! US$ 0.03/Piece Request Sample |

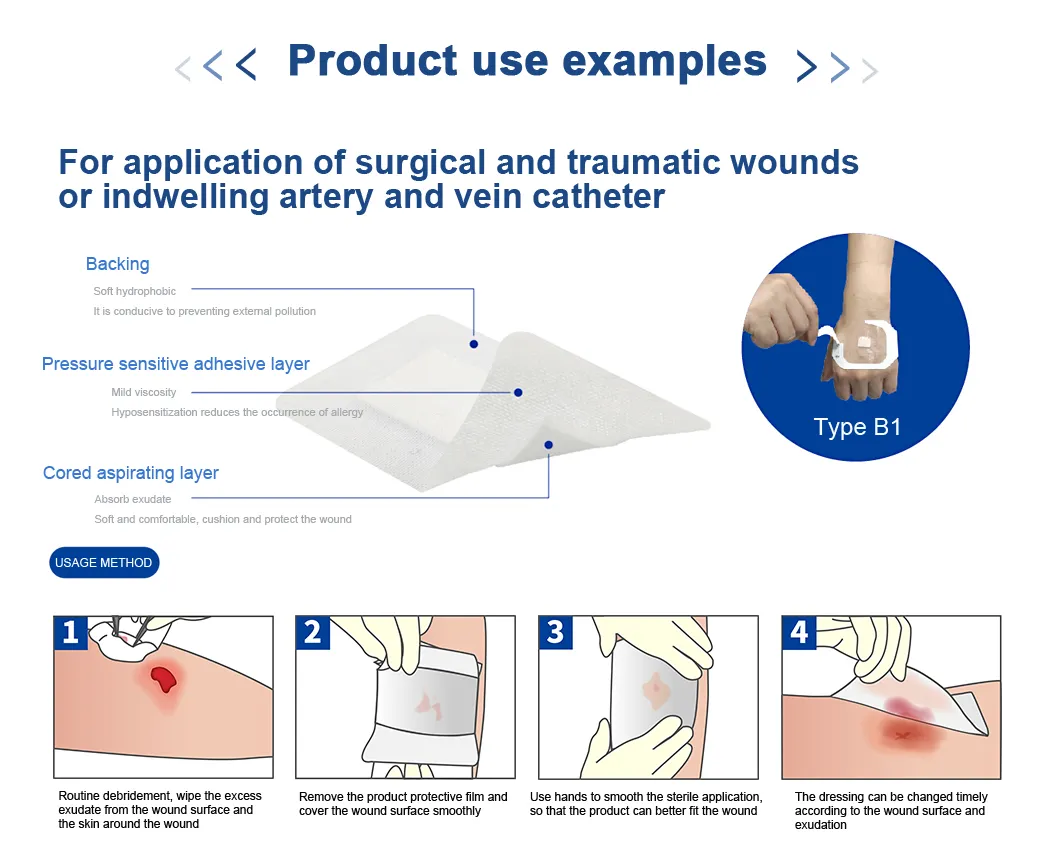
| Material | Model | Dimensions | Quantity (Sheet/box) |
|---|---|---|---|
| Non-woven | A-101 / A-103 | 6cm*7cm / 10cm*10cm | 50 / 20 |
| A-104 / A-105 | 10cm*15cm / 10cm*20cm | 20 | |
| A-106 / A-107 | 10cm*25cm / 10cm*30cm | 20 | |
| A-113 / A-114 | 9cm*20cm / 9cm*25cm | 20 | |
| PU Film | B-101 / B-103 | 6cm*7cm / 10cm*10cm | 50 / 20 |
| B-105 / B-106 | 10cm*20cm / 10cm*25cm | 20 | |
| B-110 / B-111 | 15cm*15cm / 9cm*25cm | 20 |






 Seeker Medical
Seeker Medical