 Noninvasive Pulsed Magnetic Therapy System
Noninvasive Pulsed Magnetic Therapy System
 Smart Dermatoscope with Mobile Analysis
Smart Dermatoscope with Mobile Analysis
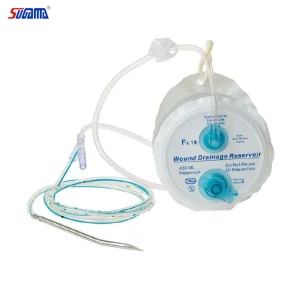 Silicone Negative Pressure Drainage System
Silicone Negative Pressure Drainage System
 Soft Breathable Non-Woven Dressing Tape
Soft Breathable Non-Woven Dressing Tape
 Polypropylene Surgical Hernia Mesh
Polypropylene Surgical Hernia Mesh
 Sterile Disposable Circumcision Device
Sterile Disposable Circumcision Device
 Sterile Medical Wooden Tongue Depressor
Sterile Medical Wooden Tongue Depressor
 Israeli 6 Inch Emergency Compression Bandage
Israeli 6 Inch Emergency Compression Bandage
In 2024, the demand for non-invasive wound closure systems, specifically Butterfly Bandages (Wound Closure Strips), has seen a 15% year-on-year increase. Hospitals and clinics in North America and Europe are shifting towards butterfly closures for minor lacerations to reduce scarring and eliminate the need for traditional sutures.
The industry is moving toward bio-compatible adhesives and hydrocolloid integration. Modern OEM/ODM factories are now focusing on antimicrobial-infused strips and flexible non-woven materials that allow skin respiration while maintaining high tensile strength.
B2B buyers, including medical wholesalers and first-aid kit assemblers, are increasingly seeking direct-to-factory partnerships in China to balance cost-efficiency with high-grade medical certifications like ISO 13485 and CE.
Choosing the right Butterfly Bandages factory requires more than just looking at the price. It involves verifying the manufacturer's technical expertise, R&D capabilities, and quality control systems. As a leading medical consumable manufacturer, we provide end-to-end solutions for global brands.
Our butterfly bandages are engineered using medical-grade adhesive technology that ensures a secure hold for up to 7 days without causing skin irritation. We utilize reinforced filaments within the tape structure to provide the necessary tension to hold wound edges together, promoting faster healing and significantly reducing the risk of hypertrophic scars.
We have realized the automatic transformation of the production process. This ensures precision in every strip, consistent adhesive weight, and a sterile environment that exceeds standard requirements.
From custom branding on the individual packaging to specific dimensions and material adjustments (e.g., waterproof vs. breathable non-woven), our R&D team works closely with your specifications.
Our products are already trusted in diverse markets worldwide. We handle the complexities of international medical exports, ensuring all documentation for CE, ISO14001, and ISO45001 is compliant for your region.
Established in May of 2004, Nanjin Seeker Medical Co., Ltd. is a professional manufacturer specialized in research, development, production of surgical sutures and surgical needles. All of our products comply with international and interior quality standards, and are approved Production license, CE, ISO13485, ISO14001, ISO45001cerificates.

We insist on the quality policy of modern techniques, excellent quality, customers satisfaction. We attach great importance to the development and application of new technology, so far, we have obtained 29 new invention patents, we have realized the automatic transformation of the production process. And we are greatly appreciated in variety of different markets throughout the world.

"All staffs in our company welcome every friend from home and abroad. Let us make more efforts for human health career."